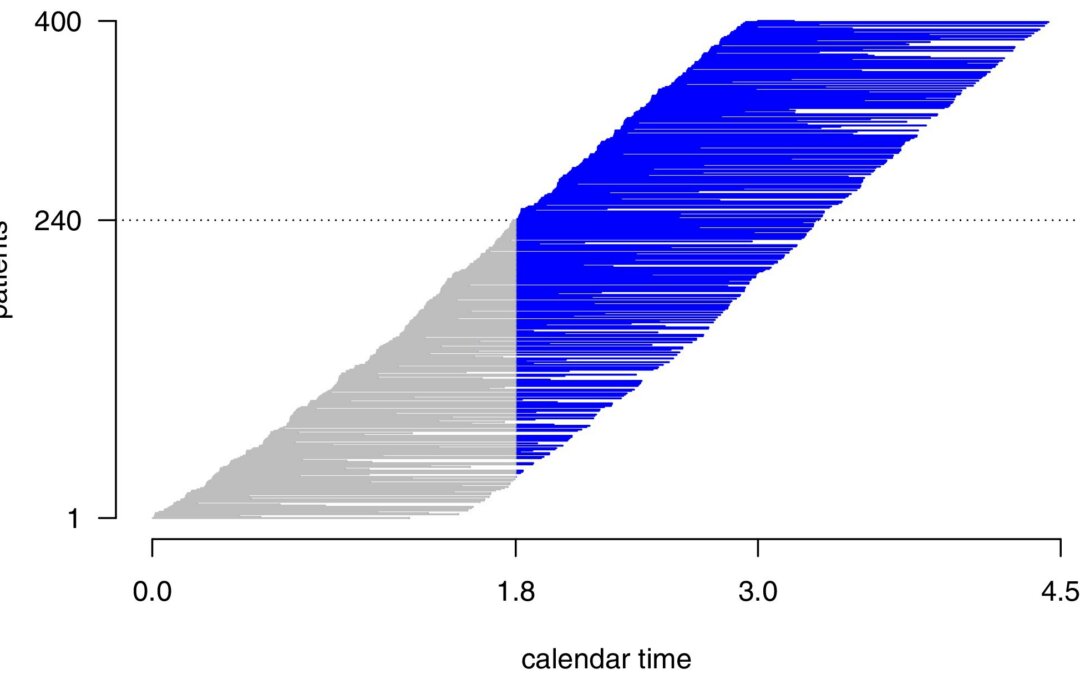
by Usha Govindarajulu | Mar 25, 2026 | Biostatistics, Blog, Usha Govindarajulu
March 25, 2026 The goal of this article was to present a methodology to design and analyze an adaptive clinical trial with sample size recalculation at interim analysis, when the restricted mean survival time (RMST) is the primary endpoint. Especially, we focus on the...

by Usha Govindarajulu | Apr 23, 2025 | Biostatistics, Blog, Usha Govindarajulu
April 23, 2025 The authors have discussed a sample size calculation for restricted mean survival time (RMST) in augmented tests. The RMST was developed as an alternative measure of survival that is non-parametric and does not reply on parametric constraints. It had...
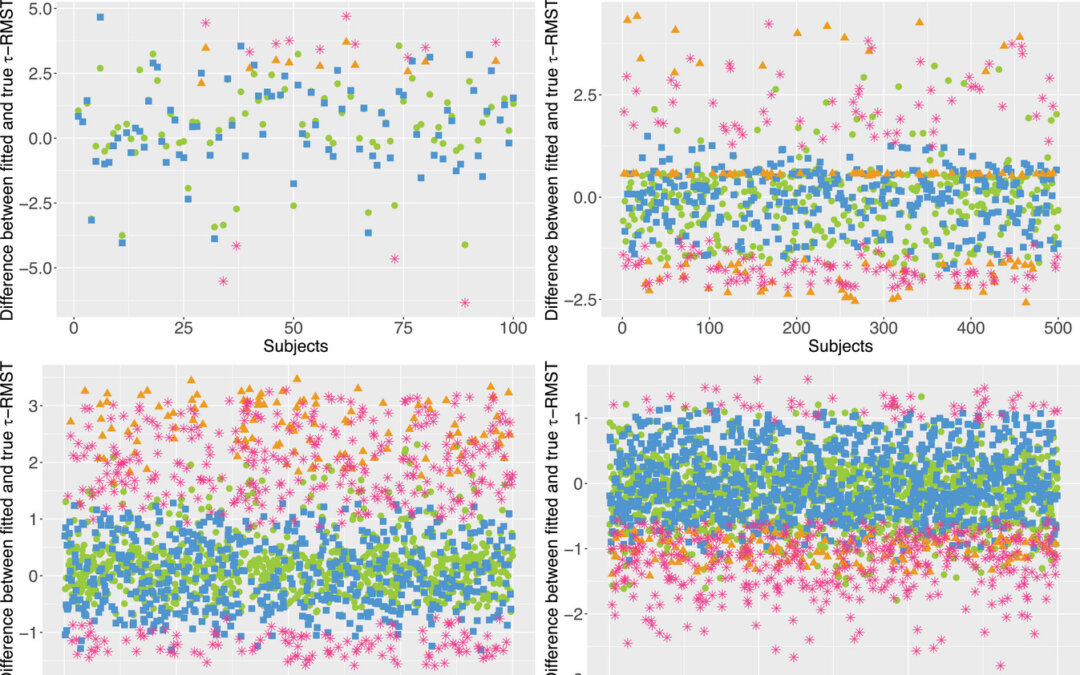
by Usha Govindarajulu | Dec 4, 2024 | Biostatistics, Blog, Usha Govindarajulu
December 4, 2024 In restricted mean survival time (RMST) analyses, tau is set at some fixed point or as stated as this paper, treated as a nonrandom, prespecified quantity, sometimes derived from the observed data (Tian, 2020). They developed tau-inflated beta...

by Usha Govindarajulu | Aug 15, 2024 | Blog
August 15, 2024 In clinical trials that use time to event endpoints, a traditional measure has been using the hazard ratio derived from a Cox proportional hazard regression, but one must satisfy this assumption. Over time, measures that have relaxed this assumption...
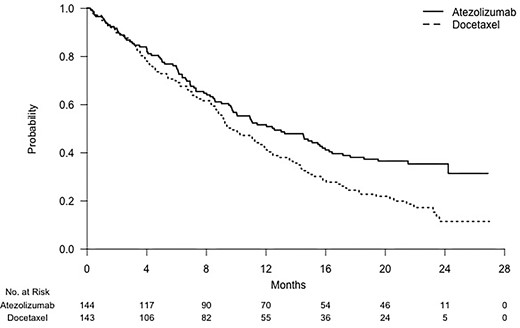
by Usha Govindarajulu | Jul 17, 2024 | Biostatistics, Blog, Usha Govindarajulu
July 17, 2024 The authors have written a new article about using average hazard (AH) as compared to restricted mean survival time (RMST) as a measure instead of hazard ratios which are instantaneous measures of effect generally obtained through estimation of a Cox...





