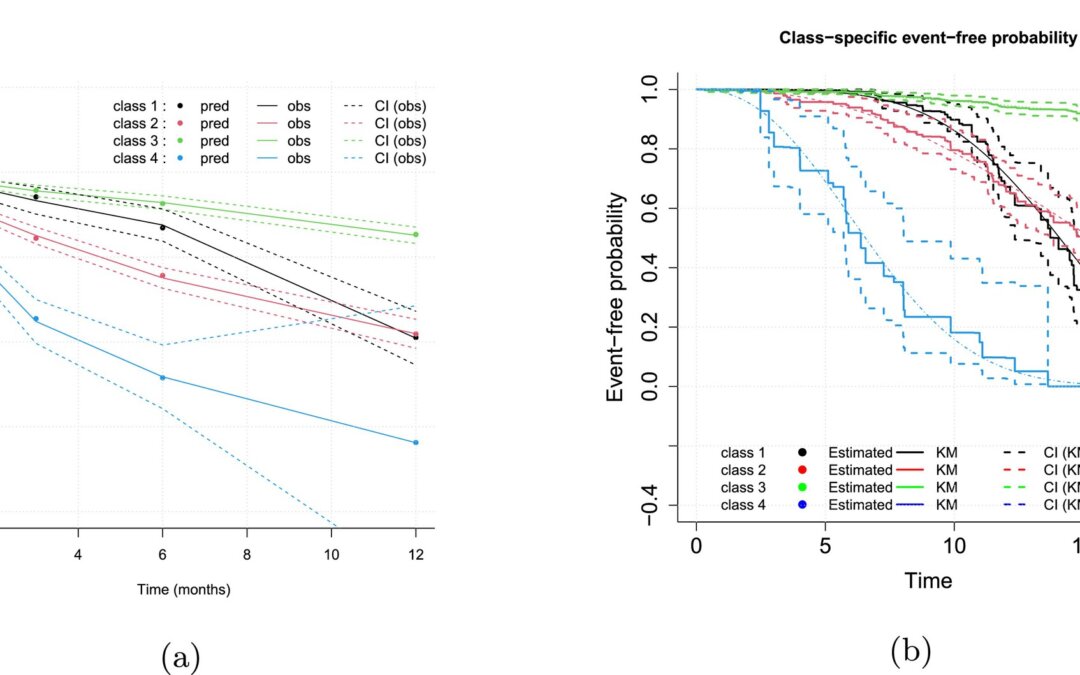
by Usha Govindarajulu | May 7, 2025 | Biostatistics, Blog, Usha Govindarajulu
May 7, 2025 The authors have presented a tutorial on joint latent class models (JLCMs), which are a statistical approach to allowing to simultaneously account for two outcomes related to disease progression, a longitudinal measure and a time-to-event measure. A linear...

by Usha Govindarajulu | Jan 15, 2025 | Biostatistics, Blog, Healtcare, Usha Govindarajulu
January 15, 2025 In this paper the authors proposed prognostic accuracy measures for recurrent event data. They did so by developing semiparametric estimators for making inferences about the prognostic accuracy measures that accommodate recurrent events. Usually a...

by Usha Govindarajulu | Jan 2, 2025 | Biostatistics, Blog, Usha Govindarajulu
January 1, 2024 The authors were interested in the average treatment effect (ATE) which reflects how the treatment affects the potential outcome. In order to estimate ATE, propensity scores have been adapted for their estimation. such as the inverse probability...
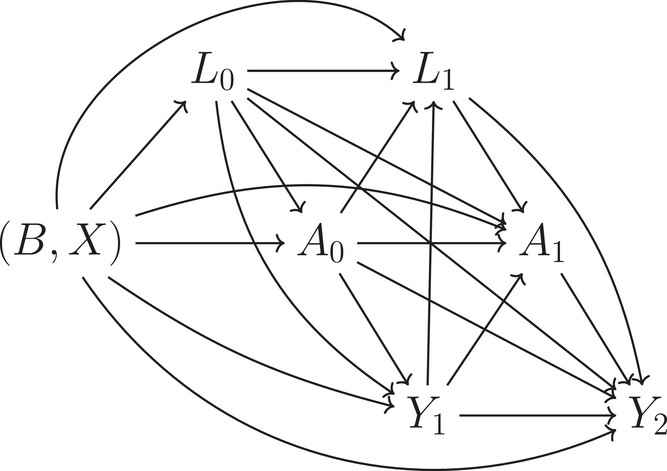
by Usha Govindarajulu | Dec 18, 2024 | Biostatistics, Blog, Usha Govindarajulu
December 18, 2024 Marginal structural models (MSMs) are often used in settings for longitudinal studies with time-varying treatment or covariates observed for each individual at several time points when trying to estimate the causal effects in a way that accounts for...
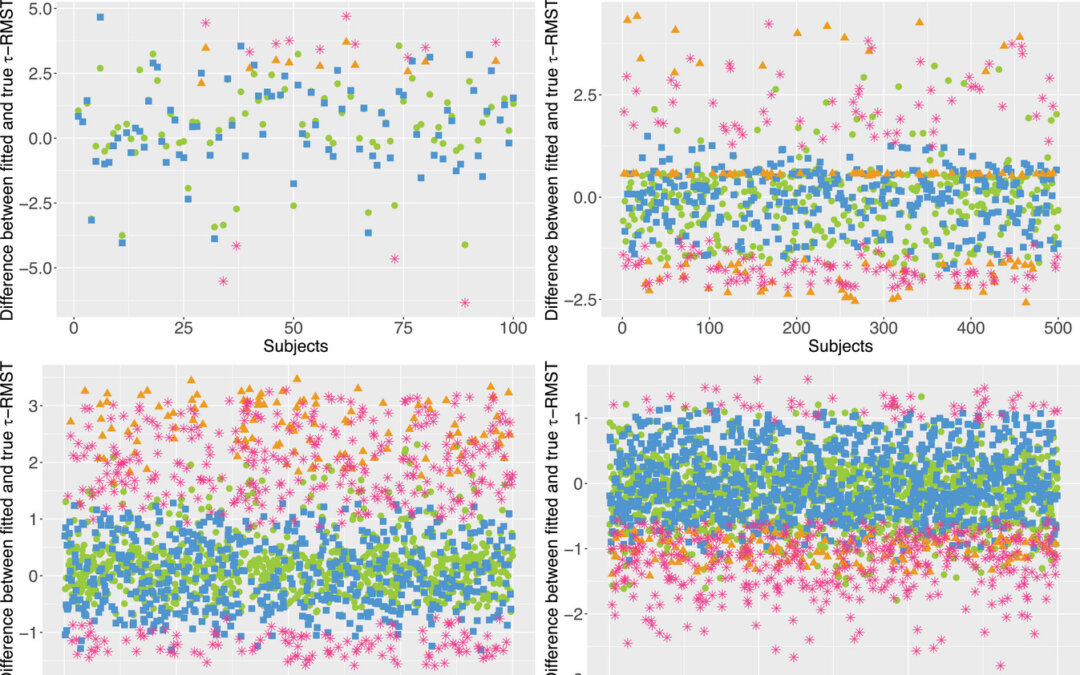
by Usha Govindarajulu | Dec 4, 2024 | Biostatistics, Blog, Usha Govindarajulu
December 4, 2024 In restricted mean survival time (RMST) analyses, tau is set at some fixed point or as stated as this paper, treated as a nonrandom, prespecified quantity, sometimes derived from the observed data (Tian, 2020). They developed tau-inflated beta...





